A young man arrives at A&E. He has recently broken up with his girlfriend and decided to kill himself. He swallowed a number of paracetamol (acetaminophen) tablets but 3 or 4 hours later decided that he did not, after all, want to commit suicide. When questioned about the number of tablets taken, he is rather uncertain, but thinks he has taken about 30. Apart from feeling nauseous, the man lacks other significant symptoms.
-
(a) Should this patient be given activated charcoal?
Show Answer
Correct answer:
No, it’s too late. Activated charcoal is only worthwhile if administered within 1 h of paracetamol ingestion.
-
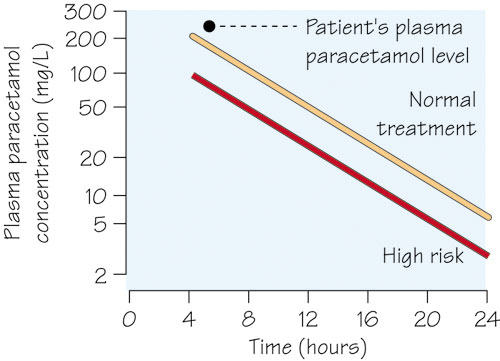
(b) Five hours after ingestion, the plasma concentration of paracetamol is found to be 230 mg/L. How does this value help in assessing the risk of liver damage in this patient?
Show Answer
Correct answer:
Providing it is at least 4 h since the ingestion of paracetamol, the plasma concentration of drug can be used to assess the likelihood of liver damage by using a plot of paracetamol concentration against time (see British National Formulary and Chapter 45). The nomogram below is based on outcome studies of many fatal and non-fatal cases of poisoning carried out before effective treatment became available. If the plasma concentration is above the normal treatment line, as it is in this patient, then the antidote is administered.
-
(c) You decide the patient is at risk of liver damage. What antidote would you administer? What test could you use to monitor liver damage caused by paracetamol?
Show Answer
Correct answer:
N-Acetylcysteine (NAC) is an effective antidote, and intravenous infusion given within 24 h protects the liver from damage. NAC is most effective if administered within 8 h but treatment continuing for up to 72 h may provide benefit. If NAC is unavailable, oral methionine is also effective but administration is difficult if the patient is vomiting.
The international normalized ratio (INR: ratio of prothrombin time to normal prothrombin time) is a sensitive measure used to monitor liver damage.
-
(d) What is the mechanism of paracetamol hepatotoxicity and how does the antidote work?
Show Answer
Correct answer:
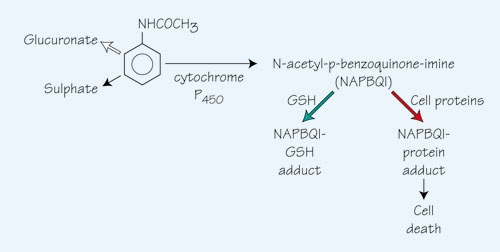
Paracetamol (acetaminophen) is N-acetyl-p-aminophenol.
Normal doses of paracetamol are metabolized to glucuronate and sulphate. However, high doses saturate these processes and P450 mixed function oxidases produce a toxic metabolite, N-acetyl-p-benzoquinone imine (NAPBQI). This may be inactivated by conjugation with glutathione (GSH) ([ ]) but toxic doses deplete the GSH stores and NAPBQI then reacts with cell proteins [
]) but toxic doses deplete the GSH stores and NAPBQI then reacts with cell proteins [ ]). This causes hepatocellular necrosis and, much more rarely, renal tubular necrosis. Regeneration of GSH requires cysteine, the availability of which can be limiting. NAC and methionine can substitute for cysteine and by increasing the synthesis of GSH they divert the reaction of NAPBQI away from cell proteins.
]). This causes hepatocellular necrosis and, much more rarely, renal tubular necrosis. Regeneration of GSH requires cysteine, the availability of which can be limiting. NAC and methionine can substitute for cysteine and by increasing the synthesis of GSH they divert the reaction of NAPBQI away from cell proteins.